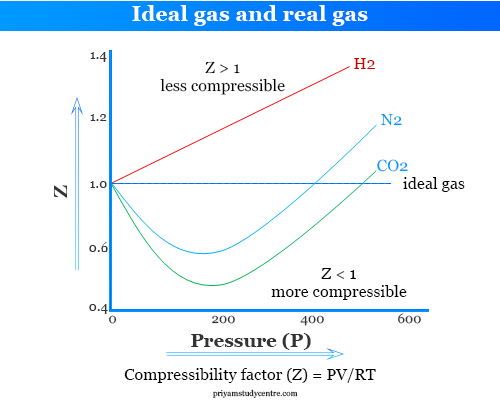
Ideal–Universal Gas Law
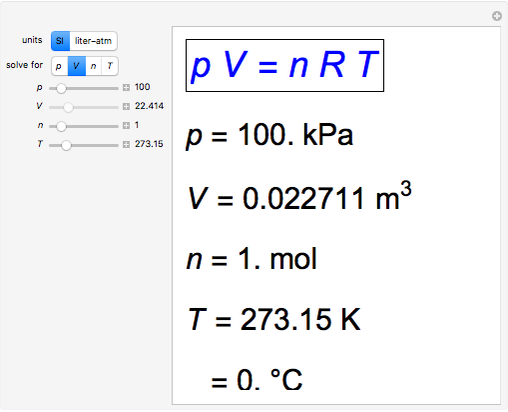
Definition: The Universal or Ideal Gas Law describes the relationship between all four properties (pressure, volume, number of moles, and temperature) as well as a gas constant called “R.” NOTE: The Ideal Gas Constant “R” has constant a value of 0.0821 L.atm/mol.K Relation: The relation between pressure (P) volume (V), number of moles (n) and…

Universal Gas Law Study Guide - Inspirit Learning Inc

Gay Lussac's Law

Hess' Law
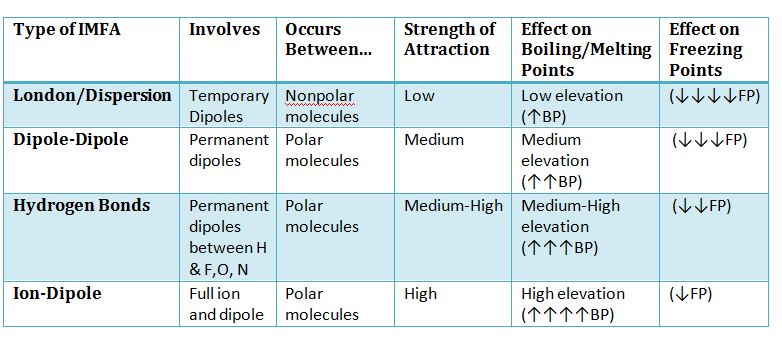
Intermolecular Forces of Attraction
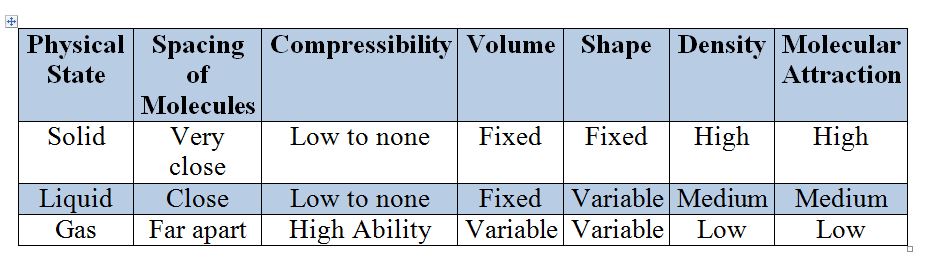
Physical States of Matter

Deriving the combined and Ideal gas Laws (part 2)

ANESTHESIA EQUIPMENT AND GAS LAW REVIEW - ppt download
SI Units

Ideal Gas Constant (R) - Universal Gas Constant

6.3: Combining the Gas Laws: The Ideal Gas Equation and the General Gas Equation - Chemistry LibreTexts

Hess' Law

Solved Write three different forms of ideal gas law using

Precipitation Reactions