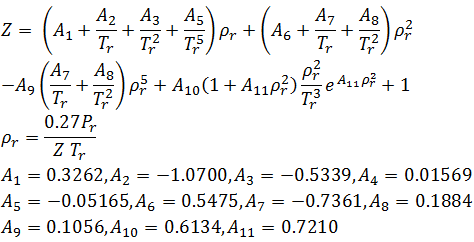Solved RT B 2. The compressiblity factor for a gas is
4.8
(683)
Write Review
More
$ 19.00
In stock
Description
Answer to Solved RT B 2. The compressiblity factor for a gas is

Non-Ideal Gas Behavior Chemistry: Atoms First

The given graph represents the variation of Z (compressibility factor = \[\dfrac{{PV}}{{nRT}}\] ) versus P, for three real gases A, B and C. Identify the only incorrect statement.

Which of the following statements is/are correct? (a) all real gases are less compressible

Real Gases Introductory Chemistry

Gas compressibility factor Z: Ideal gas vs Real gas

342 For methyl chloride at 100 C the second

Solved RT B 2. The compressiblity factor for a gas is

Solved] The Redlich-Kwong equation of state is gi

Real Gases and the Virial Equation
Related products
You may also like